Clinical Research Solutions
Understanding Bioequivalence and Product-Specific Guidances
The FDA regularly issues new and revised product-specific guidances to facilitate the availability of generic drugs and assist the generic pharmaceutical industry with identifying the most...

Clinical Research Solutions
Meet the Expert: Daniel Solorio
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...
Regulatory Sciences
Critical Issues in Drug Development: Don't Get Derailed in Developmental Efforts
Pharmaceutical companies spend massive amounts of money on drug development. Regardless of a firm’s size, the investment is substantial; the smallest error can have catastrophic consequences,...
Clinical Research Solutions
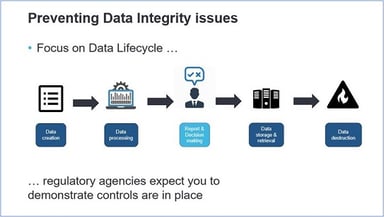
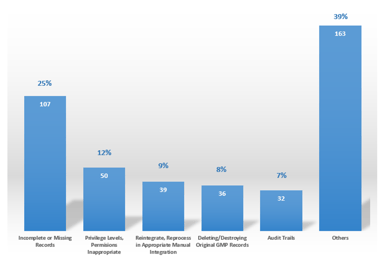
Five Steps Toward a Mature Data Integrity Culture
The corporate and quality culture has a significant effect on the maturity level of Data Integrity within a regulated company and should, therefore, be assessed and understood. To achieve the level...

Clinical Research Solutions
How to Present Scientific Data That Generates Regulatory Approvals
Today’s patients benefit from having access to the safest, most advanced pharmaceutical systems around the world. This is because of the work done by the various regulatory bodies who govern the...

Clinical Research Solutions
Meet the Expert: Hanna Edling
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...
Clinical Research Solutions
Have an FDA Submission on Your 2021 To-Do List?
At the start of every year, we all have these grand plans of everything we plan to accomplish. It is a fresh start to really get stuff done and we have a full 12 months to do it all. However,...

Clinical Research Solutions
Meet the Expert: Bram Lardée
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...